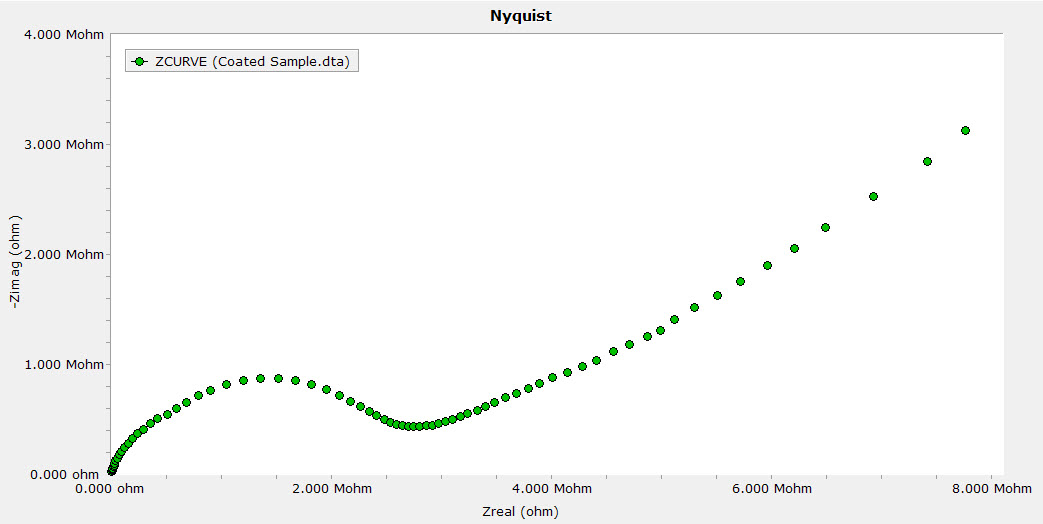
an equivalent electrical circuit, EEC) for its interpretation and analysis, fundamentally seeking a meaningful physical interpretation. After, it is fitted to a mathematical model (i.e. First, EIS data is collected through a potentiostat/galvanostat apparatus. Consequently, it is experimentally possible to determine through the EIS technique the mechanism and control that kinectics of corrosion reactions encounter. 
Corrosion has long been recognized as one of the most expensive stumbling blocks that concern many industries and government agencies, because it is a steel destructive phenomenon that occurs due to the chemical interaction with aqueous environments and takes place at the interface between metal and electrolyte producing an electrical charge transfer or ion diffusion process. However, there is no doubt that the electrochemical impedance spectroscopy (EIS) is still one of the most useful techniques around the world for metal corrosion control and its monitoring. Furthermore, its application has recently appeared to be destined in the Biotechnology field as an effective tool for rapid microbiologic diagnosis of living organism in situ. AC impedance measurements have been applied for over twenty years in electrochemistry and physics to investigate the electrical properties of conductive materials and their interfaces using an external electrical impulse (VOLTAGE, V or CURRENT, I) as driving force.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
